EctoMap
A molecular atlas of the developing ectoderm defines neural, neural crest, placode, and nonneural progenitor identity in vertebrates.
PLoS Biol. 2017 Oct 19;15(10):e2004045. doi: 10.1371/journal.pbio.2004045. [Epub ahead of print]
Plouhinec JL, Medina-Ruiz S, Borday C, Bernard E, Vert JP, Eisen MB, Harland RM, Monsoro-Burq AH.
Click here to go to EctoMap-lite.
EctoMap allows exploring spatial gene expression in the developing neurula ectoderm, in the frog X. laevis based on the transcriptome of dissected regions of the ectoderm. This online version is a light version of EctoMap, focused on the 4000 genes most differentially expressed in the ectoderm of the developing neurula. For analysis of all 31000 genes expressed during neurulation and additional features, including co-expression network analysis, please download the complete application.
Click here to view this article at PLoS Biology - download PDF.
Click here to view this article at Pubmed.
Click here to view this article on Xenbase.
Abstract
During vertebrate neurulation, the embryonic ectoderm is patterned into lineage progenitors for neural plate, neural crest, placodes and epidermis. Here, we use Xenopus laevis embryos to analyze the spatial and temporal transcriptome of distinct ectodermal domains in the course of neurulation, during the establishment of cell lineages. In order to define the transcriptome of small groups of cells from a single germ layer and to retain spatial information, dorsal and ventral ectoderm was subdivided along the anterior-posterior and medial-lateral axes by microdissections. Principal component analysis on the transcriptomes of these ectoderm fragments primarily identifies embryonic axes and temporal dynamics. This provides a genetic code to define positional information of any ectoderm sample along the anterior-posterior and dorsal-ventral axes directly from its transcriptome. In parallel, we use nonnegative matrix factorization to predict enhanced gene expression maps onto early and mid-neurula embryos, and specific signatures for each ectoderm area. The clustering of spatial and temporal datasets allowed detection of multiple biologically relevant groups (e.g., Wnt signaling, neural crest development, sensory placode specification, ciliogenesis, germ layer specification). We provide an interactive network interface, EctoMap, for exploring synexpression relationships among genes expressed in the neurula, and suggest several strategies to use this comprehensive dataset to address questions in developmental biology as well as stem cell or cancer research.
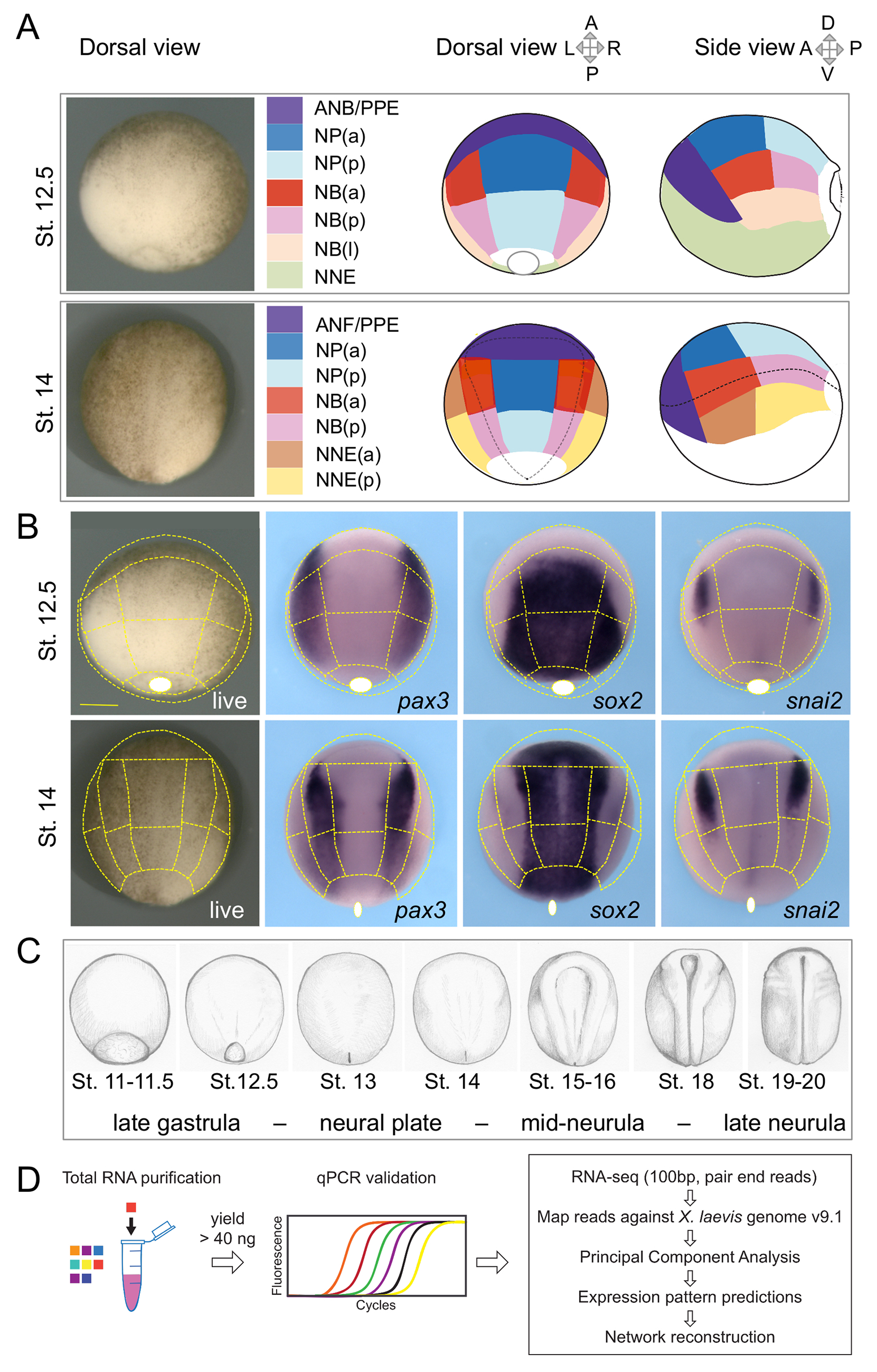
Fig 1. Ectoderm microdissections and experimental strategy. (A) Xenopus laevis neurula-stage embryos were dissected at Nieuwkoop and Faber stage (St.) 12.5 and St. 14 according to the patterns indicated. Live embryos are shown in dorsal view. The superficial ectoderm was separated from the underlying mesoderm germ layer. The tissues collected corresponded to the developing neural plate (NP, blue), medial and lateral neural border (NB, red-pink-beige), anterior neural border and preplacodal ectoderm (ANB/PPE or ANF/PPE, violet), and nonneural ectoderm (NNE, green, yellow-brown). NP, NB (at St. 12.5 and St. 14), and NNE (at St. 14 only) were further subdivided into 2 or 3 pieces: anterior (a, darker color) and posterior (p, lighter color) fragments, as indicated. For St. 12.5 NB only, a more lateral fragment was cut (l, beige), (lateral refers to medial-lateral coordinates on the dorsal view or corresponds to more ventral if using dorsal-ventral whole embryo coordinates). Each sample is associated to 1 color throughout this study (color code reference in S1 Table). (B) This dissection pattern (dotted lines) matches with reference genes expression patterns in the developing neurula ectoderm as indicated for pax3 (dorsal-lateral NP and NB), sox2 (NP and St. 14 PPE), and snail2 (premigratory neural crest within the NB). The dissection line is indicated outside the embryo whenever the dissection encompasses tissues located more ventrally than seen in dorsal view (e.g., for PPE and NNE). Similarly, the blastopore (white circle) can be indicated shifted outside the embryo if located more ventrally. (C) Whole embryos at each stage of neurulation were used for studying the time course of gene expression in neurulas. Drawing after living embryos. (D) The main steps of sample treatments and bioinformatics analysis are indicated. After total RNA purification from each individual dissected sample or whole embryo, quality was checked using quantitative PCR. Suitable samples (see text) were processed for deep transcriptome sequencing. Details of analysis workflow are indicated in Results and Materials and methods.
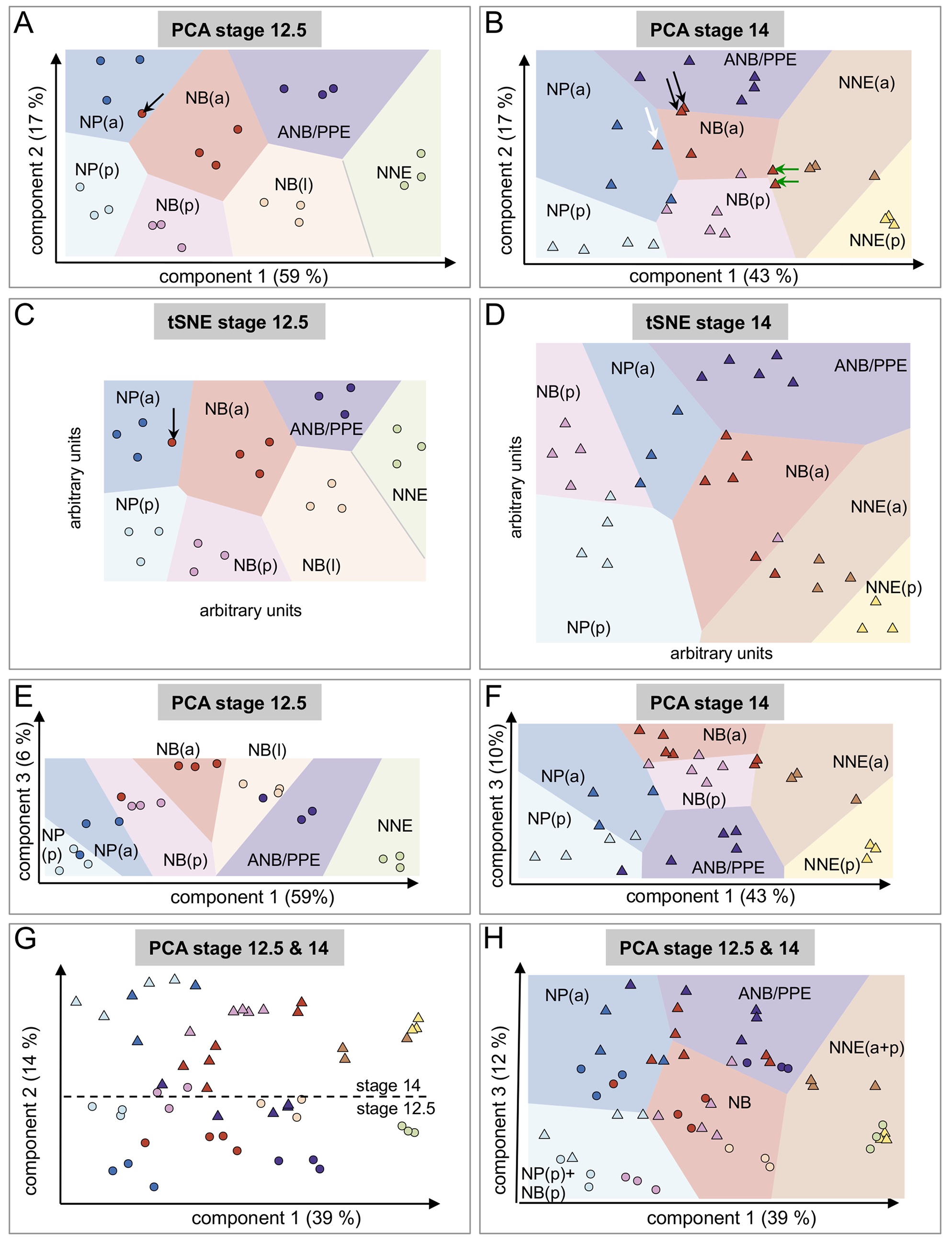
Fig 2. Unsupervised analysis of variance between samples using principal component analysis (PCA) retrieves biologically meaningful components, matching embryo polarity axes, neural border (NB) formation, and developmental stage. (A, B) At Nieuwkoop and Faber stage (St.) 12.5 (A, circles) and St. 14 (B, triangles), the first 2 PCA components segregated ectodermal samples according to their position along the dorsal-ventral (D-V) and anterior-posterior (A-P) embryonic axes, respectively. Percent of total gene expression variance captured by each component is indicated between parentheses. Sample color code is identical to Fig 1A. Lighter background colors are Voronoi diagram areas. These areas are drawn using the barycenter of samples belonging to the same dissected region. Each area indicates the regions of the PCA plane, where all points are closer to the barycenter of this dissected region than to any other barycenter. Arrows indicate anterior neural border samples falling slightly outside their intended dissected region (see text for details). (C, D) At St. 12.5 (C) and St. 14 (D), t-distributed stochastic neighbor embedding (tSNE) groups samples according to dissected regions. (E, F) At St. 12.5 (E) and St. 14 (F), neural border samples are located higher than most others along the third PCA component. This indicates that component 3 correlates with the acquisition of the NB characteristics during neurulation. (G, H) When combining St. 12.5 (circles) and St. 14 (triangles) samples together, the first component remains correlated to the D-V axis. The second component sorts out most samples according to their stage, irrespective of their position along the D-V axis (triangles separated from circles, (G). The first and third PCA components segregate most samples according to their position along the D-V and A-P axes, irrespective of their stage (circles and triangles of same color together, [H], Voronoi diagram). The posterior neural border (NBp), which is known to resemble the posterior neural plate (NPp) at St. 12.5, grouped with the NPp, while the anterior neural border (NBa)–St. 12.5, NBa–St. 14, and the lateral neural border (NBl)–St. 12.5 belong to the same Voronoi cell. As noticed above, NBa–St. 14 is more heterogeneous (red triangles).
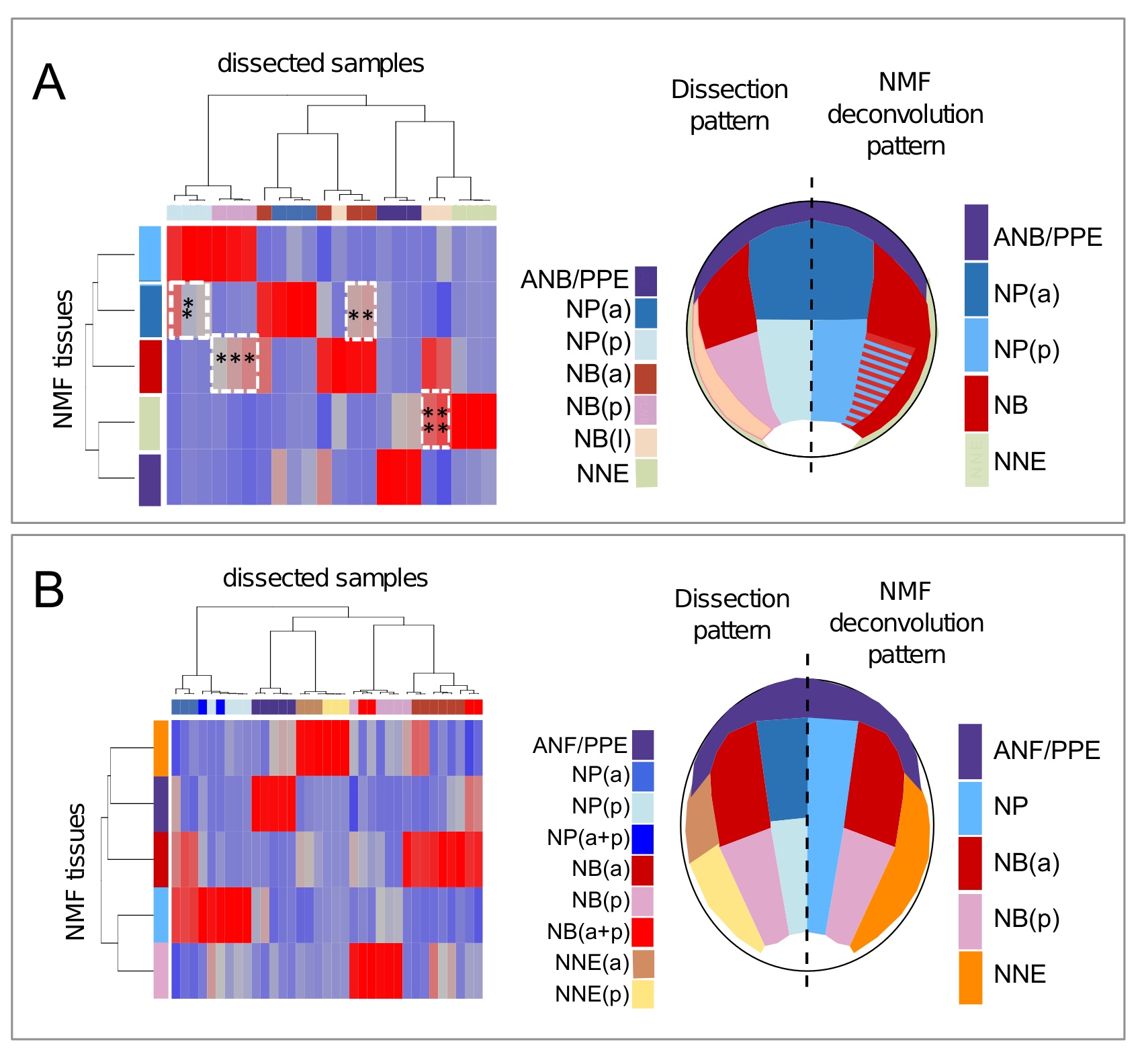
Fig 3. Nonnegative matrix factorization (NMF) efficiently deconvolutes differential expression profiles. The NMF algorithm defines a set of ectoderm tissues (NMF-tissues) at Nieuwkoop and Faber stage (St.) 12.5 (A) and St. 14 (B). Columns in the mixing matrix indicate how these tissues contribute to the initial dissected samples. Red indicates high contribution; blue indicates low/no contribution. Interesting intermediate contributions are outlined by white dotted lines; stars (*) refer to comments in text. From these contributions, dissected samples (left part of the pattern drawn on embryo) were matched with 1 of the NMF-tissues (right part of the pattern). At St. 12.5, striped pattern indicated the mixed contribution of posterior neural plate and neural border (NMF-NP(p), NMF-NB(p)) to NBp samples. Abbreviations and colors are the same as in Fig 1 and S1 Table.
Adapted with permission from PLoS Biology: Plouhinec et al. (2017). A molecular atlas of the developing ectoderm defines neural, neural crest, placode, and nonneural progenitor identity in vertebrates. PLoS Biol. 2017 Oct 19;15(10):e2004045. doi: 10.1371/journal.pbio.2004045. Copyright 2017.
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
